Site Map
© 2024 PuraCap® Pharmaceutical LLC
All Rights Reserved.
This site is only intended for residents of the United States. The products discussed on this website may have different labeling in different countries
PuraCap® Pharmaceutical LLC,
100 Somerset Corp Blvd, Ste 3000
Bridgewater, NJ 08807
Phone: 908.941.5456
Fax: 908.941.5457

Services
Research and Development
Expertise in Innovative Delivery Systems within Multiple Therapeutic Area’s
With decades of experience in drug formulation and production management (including numerous patent approvals), our highly-skilled Research and Development (R&D) team is dedicated to developing solutions that enhance the delivery system of defined molecules currently available in the global market. Our particular expertise is in soft-gelatin capsule technology and control release oral solid dosage. We are capable of developing soft-gelatin capsule solutions for current as well as future products and have a demonstrated track-record of solving formulation issues in this area. Our scientists hold several patents in this field.
PuraCap scientists are particularly interested in solving formulation challenges by new options such as tablets inside capsules, various release mechanisms in soft-gel solutions and extended release formulations. PuraCap R&D’s soft gel capsule technology is just one of several innovative modes of medication delivery we utilize to make it easier for patients to consistently comply with prescribed treatment.

Humanwell PuraCap Pharmaceuticals (Wuhan), Co., Ltd,
Manufacturing Focused on Quality & Delivery
A wide range of PuraCap products are manufactured by our affiliated manufacturing partner, Humanwell Puracap Pharmaceuticals (Wuhan) Co., Ltd, a US FDA cGMP compliant facility that complies with international regulatory standards. Our goal is to put quality first, by building quality through design and a commitment to the highest standards. We strive to exceed USFDA, cGMP standards to assure our customers the quality product they expect.
The experienced manufacturing team, led by a well-recognized technical manufacturing authority in the soft gelatin pharmaceutical capsule industry, is committed to growing our business responsibly – utilizing proven and innovative processes and procedures to produce quality products without compromise. The manufacturing team is backed by a U.S. executive team with decades of experience in FDA (Federal Drug Administration) regulated prescription drug cGMP quality system management and process validation.
The manufacturing team strives to provide cost-efficiency through manufacture at state-of-the art facilities built to USFDA specifications. Our model for success is to make use of labor as well as manufacturing efficiencies while meeting the highest quality standards. This model sets us apart from the competition. Our customers can be confident in our products—-highest quality at a competitive price. Our assurance is high quality, reliable products using the latest state of the art equipment including:
- Gelatin mixing/melting stations
- Pharmaceutical mixing/heating/cooling stations, in independent suites
- 6×10″ encapsulation machines in independent suites
- Enhanced capsule drying technologies
- In process test instrumentation
- Independent capsule inspection suites
- Independent capsule printing suites
- Independent capsule packaging suites
- Full bottling and blistering line capabilities in an independent suites
Located in the Wuhan National Bioindustry Base (Biolake) at East Lake National Innovation Demonstration Zone, learn more HERE.


Business Development
Excellence in Collaborative Business Expansion
PuraCap Pharmaceutical continuously seeks opportunities to grow and strengthen our business through acquisition, licensing, strategic alliances and other professional collaborations.
As our expertise spans many therapeutic areas within the prescription drug and Over the Counter healthcare segments, we are not limited to specific disease areas or target audiences, and remain interested in exploring a wide range of business opportunities that align with our vision of building a global pharmaceutical company renowned for cost-efficient, world-class pharmaceutical products grounded in scientific integrity.
For more information on PuraCap Business Development opportunities, please contact the Business Development team at 908.941.5456 or EMAIL.


Manufacturing A World of Wellness
Global expertise in product development; producing quality, affordable pharmaceutical and healthcare products.
Click here to join our community
homeHOME











All PuraCap Pharmaceutical prescription
brand products meet our rigorous standards
for quality, accessibility and ease of therapeutic delivery.
PuraCap Laboratories develops & markets prescription generic medications to meet patient needs for quality, efficacy and affordable products.
Our over the counter (OTC) business, Puravation Pharmaceuticals provides many top quality, OTC store brand &
private label products.
With our turn-key services, we can develop Prescription and OTC soft gelatin capsules in our state of the art facility, designed and constructed in accordance with all US FDA cGMP regulatory guidances.









November 16, 2015
South Plainfield, NJ– November 3, 2015 – PuraCap Pharmaceutical LLC
announced today the introduction of a
new product, EpiCeram-L™ Lip Care,
a unique lip care formulation with an
exclusive delivery
July 10, 2014
PuraCap™ Pharmaceutical Introduces
Unique EpiCeram® 225g Airless Pump
EpiCeram® Controlled Release Skin
Barrier Emulsion Now Offers Dosing
Flexibility with 2 Sizes.
November 13, 2015
South Plainfield, NJ–November 11, 2015 – PuraCap Pharmaceutical LLC. is pleased to announce that the US Food and Drug Administration (FDA) has completed a successful inspection of its affiliated
May 19, 2014
Max Baucus, US Ambassador to China visits Wuhan Softgel Capsule Manufacturing Plant in Wuhan China
SOUTH PLAINFIELD, N.J., May 19, 2014 /PRNewswire/ — PuraCap Pharmaceutical today announced


Site Map
© 2024 PuraCap® Pharmaceutical LLC
All Rights Reserved.
This site is only intended for residents of the United States. The products discussed on this website may have different labeling in different countries
PuraCap® Pharmaceutical LLC,
100 Somerset Corp Blvd, Ste 3000
Bridgewater, NJ 08807
Phone: 908.941.5456
Fax: 908.941.5457


Services
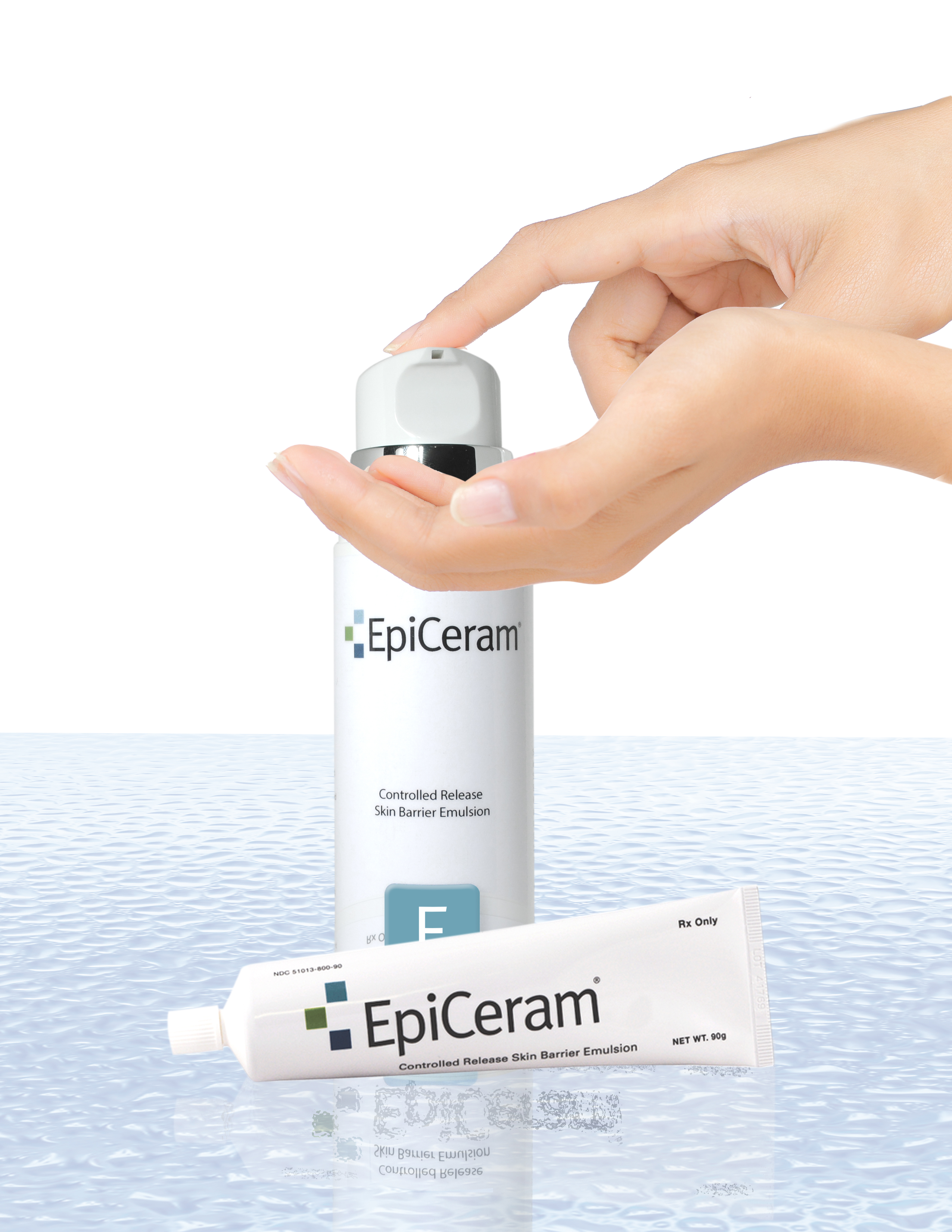
EpiCeram® is a leading prescription product that has the ability to help repair and heal the skin barrier through a unique mode-of-action different from other medications and eczema treatments. Typically, EpiCeram® is prescribed along with a topical steroid product, but EpiCeram® can also be prescribed alone. Only EpiCeram® contains the skin’s natural level of 3 essential lipids, such as ceramides, cholesterol and free fatty acids, which are reduced in patients with eczema or atopic dermatitis. EpiCeram® is steroid-free, fragrance-free, noncomedogenic1 , paraben-free, and propylene glycol-free.
EpiCeram-L™ Lip Care is the first of its kind, using a patented MultiSal™ Technology an exclusive multi-component controlled-release of important lipids, creating a moist, luxuriant feel.
EpiCeram-L™ Lip Care contains 3 essential lipids ceramides, conjugated linoleic acid (CLA) and cholesterol.
EpiCeram-L™ Lip Care is steroid-free, paraben-free, gluten-free, petrolatum-free, fragrance-free, color-free, and aloe rich to soothe lips on contact. It is ideal for the most severe, dry, cracked lips.




PuraCap Laboratories develops a wide range of prescription generic medications to meet patient needs for quality and affordability. With our own ANDA development, strategic partnerships, in-licensing agreements, we offer a wide range of high quality prescription generics.
A wide range of PuraCap products are manufactured in our affiliated manufacturing partner, Humanwell Puracap Pharmaceuticals (Wuhan) Co., Ltd, a US FDA cGMP compliant facility that comply with international regulatory standards. Our goal is to put quality first, by building quality through design and a commitment to the highest standards. We strive to exceed USFDA, cGMP standards to assure our customers the quality product they expect will be delivered.
Their experienced manufacturing team, led by a well-recognized technical manufacturing authority in the soft gelatin pharmaceutical capsule industry, is committed to growing our business responsibly – utilizing proven and innovative processes and procedures to produce quality products without compromise. Our manufacturing team is backed by a U.S. executive team with decades of experience in FDA (Federal Drug Administration) regulated prescription drug cGMP quality system management and process validation.
They strive to provide cost-efficiency through manufacture at state-of-the art facilities built to USFDA specifications. Our model for success is to make use of labor as well as manufacturing efficiencies while meeting the highest quality standards. This model sets us apart from the competition. Our customers can be confident in our products—-highest quality at a competitive price.











Our global presence in prescription brands, prescription generics, and Over the Counter (OTC) products, including private label products are growing. Our highly-skilled team meets market needs through innovation – introducing cost-efficient models to establish new product lines, utilizing global distribution networks and ensuring our products are made in world-class manufacturing facilities.
We explore ways to build expertise and expand offerings through organic growth and by seeking in-licensing opportunities, partnerships, acquisitions and other professional alliances.
- OUR VISION
To build a global pharmaceutical company renowned for affordable, world-class quality products grounded in scientific integrity.
- OUR MISSION
To enhance medication compliance and improve patient outcomes by bringing affordable, world-class quality pharmaceutical products to our customers.
- CORPORATE BROCHURE
Click to download
- OUR VISION
To build a global pharmaceutical company renowned for affordable, world-class quality products grounded in scientific integrity.
- OUR MISSION
To enhance medication compliance and improve patient outcomes by bringing affordable, world-class quality pharmaceutical products to our customers.
- CORPORATE BROCHURE
Click to download
Dahai Guo
Elise Klein
Mark Bolling
Founder and CEO of
PuraCap Pharmaceutical, LLC
General Manager,
Branded Rx Sales
Vice President,
Sales & Marketing, OTC Business
Xiaofeng Meng, PhD
Sean Weeks
Vice President
Vice President,
Global Manufacturing
Mahboob Rahman
Sr. Director R&D
Adam Feng



Over the Counter-
Branded & Private Label Products
PuraCap Pharmaceutical provides many high quality, over the counter (OTC) store brand-private label products that you frequently find at many of retailers across the USA.
View the PuraCap product list.
(All softgel OTC products are manufactured at our affiliate company Humanwell PuraCap Pharmaceuticals (Wuhan) Co., Ltd. located at Wuhan, China.)
Generic Prescription Products
PuraCap Laboratories develops a wide range of medications to meet patient needs for quality and affordability. With our own ANDA development, strategic partnerships, in-licensing agreements, we offer a wide range of high quality products. For more information please contact us through the link below.
This is a headline, click to change the text.
Prescription Brand Products
All PuraCap Pharmaceutical prescription brand products meet our rigorous standards for quality, accessibility and ease of therapeutic delivery.
Find out more about
EpiCeram® Skin Barrier Emulsion
(EpiCeram is manufactured at our contract manufacturing organization in the US.).
Research and Development
Expertise in Innovative Delivery Systems within Multiple Therapeutic Area’s
With decades of experience in drug formulation and production management (including numerous patent approvals), our highly-skilled Research and Development (R&D) team is dedicated to developing solutions that enhance the delivery system of defined molecules currently available in the global market. Our particular expertise is in soft-gelatin capsule technology and control release oral solid dosage. We are capable of developing soft-gelatin capsule solutions for current as well as future products and have a demonstrated track-record of solving formulation issues in this area. Our scientists hold several patents in this field.
PuraCap scientists are particularly interested in solving formulation challenges by new options such as tablets inside capsules, various release mechanisms in soft-gel solutions and extended release formulations. PuraCap R&D’s soft gel capsule technology is just one of several innovative modes of medication delivery we utilize to make it easier for patients to consistently comply with prescribed treatment.

Humanwell PuraCap Pharmaceuticals (Wuhan), Co., Ltd,
Manufacturing
Humanwell PuraCap Pharmaceuticals (Wuhan), Co., Ltd,
Manufacturing Focused on Quality & Delivery
A wide range of PuraCap products are manufactured by our affiliated manufacturing partner, Humanwell Puracap Pharmaceuticals (Wuhan) Co., Ltd, a US FDA cGMP compliant facility that complies with international regulatory standards. Our goal is to put quality first, by building quality through design and a commitment to the highest standards. We strive to exceed USFDA, cGMP standards to assure our customers the quality product they expect.
The experienced manufacturing team, led by a well-recognized technical manufacturing authority in the soft gelatin pharmaceutical capsule industry, is committed to growing our business responsibly – utilizing proven and innovative processes and procedures to produce quality products without compromise. The manufacturing team is backed by a U.S. executive team with decades of experience in FDA (Federal Drug Administration) regulated prescription drug cGMP quality system management and process validation.
The manufacturing team strives to provide cost-efficiency through manufacture at state-of-the art facilities built to USFDA specifications. Our model for success is to make use of labor as well as manufacturing efficiencies while meeting the highest quality standards. This model sets us apart from the competition. Our customers can be confident in our products—-highest quality at a competitive price. Our assurance is high quality, reliable products using the latest state of the art equipment including:
- Gelatin mixing/melting stations
- Pharmaceutical mixing/heating/cooling stations, in independent suites
- 6×10″ encapsulation machines in independent suites
- Enhanced capsule drying technologies
- In process test instrumentation
- Independent capsule inspection suites
- Independent capsule printing suites
- Independent capsule packaging suites
- Full bottling and blistering line capabilities in an independent suites
Located in the Wuhan National Bioindustry Base (Biolake) at East Lake National Innovation Demonstration Zone, learn more HERE.


Business Development
Excellence in Collaborative Business Expansion
PuraCap Pharmaceutical continuously seeks opportunities to grow and strengthen our business through acquisition, licensing, strategic alliances and other professional collaborations.
As our expertise spans many therapeutic areas within the prescription drug and Over the Counter healthcare segments, we are not limited to specific disease areas or target audiences, and remain interested in exploring a wide range of business opportunities that align with our vision of building a global pharmaceutical company renowned for cost-efficient, world-class pharmaceutical products grounded in scientific integrity.
For more information on PuraCap Business Development opportunities, please contact the Business Development team at 908.941.5456 or EMAIL.